Mainly, drug substances are the chemical entities that intend to go through chemical changes or degradation. Molecular changes and the presence of different functional groups cause it. Degradation starts due to several factors such as temperature, moisture, pressure, etc. A degradation reaction can also occur in organic and aqueous solvents. The drug substances undergo various pathways that cause the degradation of the product. Various reactions promote drug degradation of pharmaceutical products. Among them, Hydrolysis and Oxidation are the two reactions that undergo further drug degradation.

Hydrolysis of Procaine is primarily controlled by this reaction. In this chemical reaction, it forms 4-aminobenzoic acid and dimethylaminoethanol. Ionization of the molecule influences the reaction here.


Sulfacetamide hydrolyzes to produce acetic acid and sulphanilamide. Sulfanilamide undergoes oxidation to yield 4,4’- azobenzenedisulfonamide. On further oxidization, it yields 4,4’-azoxybenzenedisulfonamide on exposure to light. This reaction shows the formation of the reddish-brown color from yellow color.

Desethylene norfloxacin under the presence of light father hydrolyzes and is converted to N-acetyinorfloxacin.
H2A DHA+ 2e- + 2H+
Dehydroascorbic acid is converted to diketogulonic acid in an alkaline solution with the involvement of the process of hydrolysis.

The dextrorotatory form of oxidized morphine is popularly marketed as noxydimorphine or pseudomorphine.
The levorotatory form of oxidized morphine is popularly marketed as morphine-N-oxide.


Hydrolysis and oxidation reaction also causes the degradation of pharmaceutical products. Not only that but several reactions can cause the degradation of drugs. The reactions such Decarboxylation, Elimination, Isomerisation, Dimerization, Epimerization, Dehydration, Dehydrogenation, and Dehalogenation.
Hydrolysis
Hydrolysis involves the reaction of the chemical compounds with water that produces two or more products. The bond on the chemical compound is formed by the addition of water. In an aqueous solution and liquid dosage form, the hydrolytic reaction is the most common factor. A substance that contains ester, amides, imides, carbamates, lactones, nitriles, and carbohydrate groups will be destabilized by this substance. In this case, the pH of the medium plays an essential role. There are also varieties of drugs susceptible to acid as well as alkaline hydrolysis. The drugs that change by hydrolysis are aspirin, paracetamol, sulfacetamide, indomethacin, procaine, digoxin, riboflavin, benzodiazepines, penicillins, lincomycin, chloramphenicol, and cephalosporins.Hydrolysis of Ester
Water or OH- ions attack the ester group nucleophilically during the hydrolysis of ester. It undergoes a second-order reaction and is one of the most important reactions of hydrolysis. On acidic hydrolysis of ester, it yields carboxylic acid and alcohol whereas basic hydrolysis of ester yields carboxylate salt and alcohol. For example, Aspirin or Acetylsalicylic acid, and procaine. In the hydrolysis of Aspirin, it produces salicylic acid and acetic acid. On the other hand, this reaction is completely accelerated by temperature.Hydrolysis of Procaine is primarily controlled by this reaction. In this chemical reaction, it forms 4-aminobenzoic acid and dimethylaminoethanol. Ionization of the molecule influences the reaction here.
Hydrolysis of amides
It contains compounds that have amide bonds. In comparison to ester bonds, amide bonds are less susceptible to hydrolysis. Since this bond is electrophilic, the carbonyl carbon of the amide has a lower electrophilicity. In the reaction, the carbon-nitrogen bond breaks down and yields COOH, NH3, or amine. For example, Paracetamol, and Sulfacetamide. Thehydrolysis of Paracetamol leads to the formation of 4-aminophenol and acetic acid.Sulfacetamide hydrolyzes to produce acetic acid and sulphanilamide. Sulfanilamide undergoes oxidation to yield 4,4’- azobenzenedisulfonamide. On further oxidization, it yields 4,4’-azoxybenzenedisulfonamide on exposure to light. This reaction shows the formation of the reddish-brown color from yellow color.
Hydrolysis by Ring Opening
The hydrolysis of the drug molecules occurs by the cleavage of C-N bond.Riboflavin
Riboflavin is a water-soluble compound popularly marketed as vitamin B12. Riboflavin is hydrolyzed in a basic medium converting it into two compounds. The dextrorotatory part 1,2-dihydro-6,7-dimethyl-2-keto-I-D-ribityl-quinoxaline-3-carboxylic acid and levorotatory part with the IUPAC name 6,7-dimethyl-4-D-ribityl-2,3-dioxo-1,2,3,4-tetrahydro quinoxaline. The reduction of riboflavin absorption at 445 nm is accompanied by the degradation reaction, which is enhanced by increasing the temperature.Norfloxacin
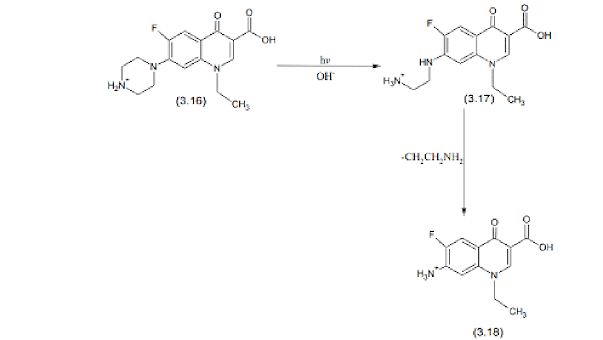
Norfloxacin is a fluoroquinolone antibiotic derivative used for solving digestive system-related problems. Norfloxacin undergoes hydrolysis and breaks the piperazine ring cleavage in an alkaline medium forming diethylene norfloxacin.Desethylene norfloxacin under the presence of light father hydrolyzes and is converted to N-acetyinorfloxacin.
Oxidation
Oxidation is the process by which an element or compound gains oxygen or loses an electron from its outermost shell. An oxidizing agent or oxygen initiates the oxidation process. It has been noted that drugs are easily oxidized in contamination with air while manufacturing, packing, and storage. The pH of the medium is capable to affect the rate of oxidation which results in ionization and change in the redox potential of the compound. Many compounds undergo the process of oxidation. These compounds include ascorbic acid, morphine, phenols, etc.Oxidation of Ascorbic acid
Ascorbic acid popularly marketed as vitamin C is a highly complex organic molecule that is soluble in water. The ascorbic acid molecule undergoes oxidation, changing its chemical structure and property. Ascorbic acid can be converted to dehydroascorbic acid during this oxidation step.H2A DHA+ 2e- + 2H+
Dehydroascorbic acid is converted to diketogulonic acid in an alkaline solution with the involvement of the process of hydrolysis.
Oxidation of morphine
Morphine is a natural tranquilizer obtained from the plant opium. It is wildly used as a painkiller after any major surgery. Morphine is oxidized in an aqueous solution by air and light. After oxidation morphine is converted into two different compounds. Morphine is oxidized to Pseudomorphine and Morphine-N-Oxide in an aqueous solution.The dextrorotatory form of oxidized morphine is popularly marketed as noxydimorphine or pseudomorphine.
The levorotatory form of oxidized morphine is popularly marketed as morphine-N-oxide.
Oxidation of phenols
Phenol is a white, crystalline solid, aromatic, water-soluble, and volatile compound used for both household purposes and in the pharmaceutical industry for synthesis purposes. Phenol has a resonating structure. The hydroxyl group of the phenol moiety is capable of donating an electron and initiating the process of oxidation. A radical is formed as a result of the proton being removed from the atom. Phenol deprotonation occurs at higher pH levels. The phenolate anion catalyzes the auto-oxidation process in a considerable way. The phenolate anion is a powerful nucleophile that can react with electrophilic species on both ends of the electron chain.Hydrolysis and oxidation reaction also causes the degradation of pharmaceutical products. Not only that but several reactions can cause the degradation of drugs. The reactions such Decarboxylation, Elimination, Isomerisation, Dimerization, Epimerization, Dehydration, Dehydrogenation, and Dehalogenation.
Get subject wise printable pdf notesView Here



No comments:
Post a Comment
Please don't spam. Comments having links would not be published.